Abstract
The currently recommended first-line treatments of erectile dysfunction (ED), phosphodiesterase type 5 inhibitors (PDE5i), for example sildenafil, are efficacious in many patients with ED of vascular origin, but this therapy is insufficient in approximately 30-40% of men with ED where there is also a neuronal affection. There is a demand of novel approaches to treat the condition. We review the possibility of modulating the dopaminergic pathways to improve erectile function. Dopamine D1 (D1 , D5 )- and D2 (D2 -D4 )-like receptors in the paraventricular area, the medial pre-optic area, the spinal cord, and in the erectile tissue are involved in erection, and several agonists developed for the treatment of Parkinson’s disease are associated with increased libido. A therapeutic window for the treatment of ED was found by sublingual administration of the general dopamine receptor agonist apomorphine, but it failed mainly due to less efficacy on erectile function compared with PDE5i. To avoid the dose-limiting side effects mediated by D2 receptors, nausea and emesis, dopamine D4 receptor agonists were developed, and they induce erection in rodents, but these drugs were never introduced clinically. The β-lactamase inhibitor clavulanic acid increases dopamine and serotonin and was found to increase sexual arousal and erections, but the dose-response curve is bell-shaped. Bupropion has selectivity for inhibition of the dopamine reuptake transporter and can be used to alleviate sexual symptoms caused by other antidepressant medication, hence providing an interesting approach to treat ED. In summary, modulation of the dopaminergic pathways provides a possibility to improve the treatment of ED.
- PMID: 27541930
- DOI: 10.1111/bcpt.12653
Erection is a hemodynamic event where vasodilatation of intracavernous and helicine arteries is followed by increased arterial blood inflow to the corpora cavernosa. Erection involves different central and peripheral neural and/or humoral mechanisms 1. Central neurotransmitters and neuropeptides can either facilitate, for example dopamine (DA), or inhibit (e.g. opioid peptides) penile erection by acting in several brain areas. Serotonin can exert both facilitatory and inhibitory effects, depending on the receptor subtype involved 2.
Peripheral neurotransmitters released from sympathetic (noradrenaline, ATP) and parasympathetic (acetylcholine, nitric oxide, vasoactive intestinal peptide) nerves entering the corpora cavernosa, corpus spongiosum and glans penis regulate blood flow during erection and detumescence 1, 3, 4. In metabolic syndrome and diabetes, neuropathy as well as an imbalance between peripheral contractile and relaxant factors in the erectile tissue can contribute to erectile dysfunction (ED) 5.
Current guidelines recommend phosphodiesterase type 5 inhibitors (PDE5i), sildenafil, vardenafil, tadalafil and avanafil, as the first‐line treatment of ED; however, approximately 30–40% of men with ED do not respond to PDE5 inhibitor therapy 6, 7. Patients suffering from neurological damage, diabetes mellitus or severe vascular disease may be resistant to PDE5 inhibitors 8. In addition to these diseases, medications of central nervous system (CNS) disorders, like antidepressants, antipsychotics and anxiolytics, have also a negative impact on the erectile function 5. Antidepressants, such as selective serotonin reuptake inhibitors (SSRIs) and venlafaxine, can negatively affect the male sexual function (desire/arousal–excitement–orgasm). Other antidepressants, like bupropion, nefazodone and mirtazapine, also affect sexual function, although the incidence of sexual dysfunction is lower, compared with SSRI 9. In addition to mood disorders, antidepressants are also used to treat neuropathic pain; therefore, sexual dysfunction induced by antidepressants affects a wider population. In some clinical trials, sildenafil corrected ED induced by antidepressant medication; however, due to an increasing number of non‐responders to PDE5 inhibitors, there is a demand for novel approaches to the treatment of ED. An approach would be to target not only the peripheral pathways but also the central pathways of importance for erectile function. As mentioned, a series of neurotransmitters are involved in erectile function both at central and peripheral levels and a series of recent reviews have addressed the regulation in detail 1, 2 and clinical studies related to sexual dysfunction and monoamines 10. A comprehensive review which covers some of the most recent drugs under development for ED can also be recommended 11. For the treatment of Parkinson’s disease, several drugs with dopaminergic effect were developed, and one of these drugs the general dopamine receptor agonist, apomorphine, was found to induce erection 12–15. Therefore, the focus of the present MiniReview is to consider the possibility of modulating the dopaminergic pathways to improve erectile function.
Dopaminergic Pathways Involved in Erectile Function
Central pathways
The central pathways involved in the control of erectile function include several brain areas such as the medial pre‐optic area (MPOA), the paraventricular nucleus (PVN) of hypothalamus, the ventral tegmental area, the hippocampus, the amygdala, the bed nucleus of the stria terminalis, the nucleus accumbens, the medulla oblongata and the spinal cord 2, where the PVN of hypothalamus and the ventral tegmental area are particularly important 2, 15. A series of neurotransmitters are involved in the central regulation of erection and they facilitate erectile function (dopamine, nitric oxide, glutamate, acetylcholine, oxytocin, hexarelin peptide, ACTH, MSH and pro‐VGF), inhibit erectile function (e.g. noradrenaline, enkephalins, GABA and endocannabinoids) or in case of serotonin both facilitate and inhibit erectile function 2. Dopamine is the main neurotransmitter in the CNS and facilitates sexual motivation, copulation and genital reflexes 1, 16. Dopamine, thought to be of importance for erectile function, is localized in the MPOA and the PVN of the hypothalamus and the nucleus accumbens. These three areas receive dopaminergic innervation from the incertohypothalamic system 17. In the PVN, dopamine leads to the activation of oxytocinergic neurons probably by increasing intracellular calcium followed by the activation of neuronal nitric oxide synthase (nNOS). Nitric oxide through a cyclic GMP‐independent pathway probably nitrosylation is thought to lead to the activation of the oxytocinergic neurons (fig. 1). Nitric oxide is formed by nNOS and castration and also exogenous testosterone, respectively, down‐and up‐regulate the expression of nNOS in the PVN 2.
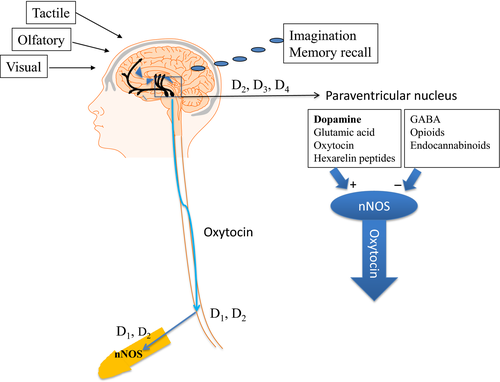
Figure 1
Furthermore, dopaminergic neurons have been identified that travel from the caudal hypothalamus to innervate the autonomic and somatic nuclei in the lumbosacral spinal cord 18, 19. Thus, dopamine can be expected to participate in the regulation of both the autonomic and somatic components of the penile reflexes.
The PVN oxytocinergic neurons project to the neurohypophysis and other brain areas, but also to medulla oblongata and the spinal cord, where it leads to the activation of the pro‐erectile lumbo‐sacral parasympathetic neurons innervating genitalia.
Spinal pathways
Apparently, a spinal dopaminergic pathway is also of importance for erectile function. Fibres immunoreactive to dopamine are present in the thoracolumbar sympathetic chain and also in the lumbosacral parasympathetic nucleus 20, 21. Moreover, a strong dopamine D2 receptor expression was found in the lumbosacral parasympathetic neurons 22. Further support for a dopaminergic pathway at spinal level comes from the observations that in rats with spinal cord lesions at the thoracic T8 segment, the dopaminergic agonist apomorphine infused systemically is able to induce erection in rats by a mechanism antagonized by dopamine D2 receptor antagonist (haloperidol, sulpiride) and facilitated by a dopamine D1 receptor antagonist (SCH23390) 23. These observations were also supported by the observations that intrathecal administration of apomorphine induces spontaneous erectile responses 24.
Peripheral pathways
Penile erection is initiated by inhibition of the sympathetic nerves and activation of parasympathetic pelvic nerves leading to penile arterial dilatation and relaxation of the erectile smooth muscle cells of the corpora cavernosa. This allows blood filling of the cavernous sinusoids and restriction of the venous out‐flow, with entrapment of pressurized blood in the corpora cavernosa. Release of nitric oxide from the parasympathetic nerves plays an important role for the relaxation, and the increase in blood inflow to penis during the erection also stimulates the endothelial cell layer to release nitric oxide and other endothelium‐dependent vasodilators contributing to maintain erection 25–27.
In contrast to the role of dopamine in the CNS, the role of dopamine in the peripheral erectile tissue is less clear. Infusion of dopamine induces erection in cats 28, and an antagonist of dopamine D1 receptors, SCH23390, markedly inhibits the increase in intracavernosal pressure induced by stimulating the cavernous nerve in rats 29. Dopamine D1 and D2 receptors are expressed in erectile tissue from rats and man 30, 31, and patch clamp of isolated smooth muscle cells from corpus cavernosum suggested that dopamine and dopaminergic agonists induce relaxation by opening of large‐conductance calcium‐activated potassium channels 32. There is evidence for the expression of the dopamine transporter in the endothelial cells of systemic arteries 33, 34, but this has still not been examined in endothelial cells of erectile tissue. Despite dopamine can be formed in sympathetic nerve terminals, the source of dopamine in erectile tissue remains to be clarified, and also whether the dopamine involved in erection derives from the endothelium or other structures in the erectile tissue.
In summary, dopaminergic pathways in CNS and in the lumbosacral part of the spinal cord are involved in the erectile function, and in the erectile tissue there is probably also a dopaminergic pathway. This suggests that modulation of the dopaminergic pathways can aim at all three levels or hydrophilic drugs targeting only the peripheral dopaminergic pathways can be attempted.
Pharmacological Modulation of the Dopaminergic Pathways to Improve Erectile Function
Dopamine receptor agonists
The first drug with dopaminergic effect found to induce erection was the general dopaminergic receptor agonist, apomorphine 12, and most of the evidence related to pro‐erectile effects has been obtained for dopaminergic receptor agonists. There are five main subtypes of dopamine receptors D1–D5 grouped as D1‐like (D1, D5), and D2‐like (D2, D3 and D4) receptors. Binding affinities are described for the dopamine receptor agonists in table 1, although that does not necessarily correspond to the functional effects of the drugs on the respective receptors. Some of the drugs are only partial agonists at the receptors meaning that high concentrations may antagonize the endogenous ligand dopamine. Moreover, homo‐ and heterodimers of dopamine receptors have been described, and that may also change both affinity and effect of the respective agonists 35.
| D1 GαS cAMP↑, Ca2+↑ | D2 Gi/Go cAMP↓, K+↑ | D3 Gi/Go cAMP↓, K+↑ | D4 Gi/Go cAMP↓, K+↑ | D5 GαS cAMP↑ | |
|---|---|---|---|---|---|
| Dopamine | 4.3–5.2 | 4.7–7.2 | 6.4–7.3 | 7.6 | 6.6 |
| Apomorphine | 5.3–6.2 | 7.6 (PA) | 6.1–7.6 | 8.4 (PA) | 6.4–7.8 (PA) |
| Bromocriptine | 6.2 (PA) | 7.3 | 7.1 (PA) | – | 6.3 (PA) |
| Fenoldopam | 6.5–7.9 | – | – | 6.5 | – |
| Cabergoline | 6.7 | 9.0–9.2 (PA) | 9.1 | 7.3 (PA) | 7.7 |
| Pergolide | 5.9–6.5 | 7.5–7.6 | 8.3 | 7.2 | 6.0–7.5 |
| Ropinirole | – | 8.1 | 7.7 | – | – |
| Rotigotine | 5.6 | 10.2 | 8.4 | 7.3 | 6.0 |
| Lisuride | 7.2 | 9.2–9.5 | 9.3 (PA) | 8.3 | – |
| Quinpirole | – | 4.9–7.7 | 6.4–8.0 | 7.5 | – |
| Quinelorane | – | 5.5–5.7 | 7.2–7.4 | – | – |
| PD168,077 | – | – | – | 8.8 (PA) | – |
| ABT 724 | – | <5 | – | 7.2 (PA) | – |
| ABT 670 | – | <5 | – | 5.8 (PA) | – |
- The table has been constructed based on the information on binding to human dopamine receptors available in the database: www.guidetopharmacology.org owned by the International Union of Pharmacology (IUPHAR) and British Pharmacological Society, where the references providing the original measurements can also be accessed. When a range is given, it reflects binding to subpopulations of the receptor subtype. Regarding ABT 724, the Ki values are from the original work 57. Some of the agonists have been characterized to be partial agonists (PA) on the receptors. The G proteins and signal transduction pathways only cover the classical pathways. For a detailed description, please see text 35.
Dopamine receptors are considered to signal through G protein‐dependent cellular pathways, where D1‐like receptors (D1 and D5) couple to GαS protein and on activation stimulate adenylate cyclase and increase cyclic AMP, while D2‐like receptors (D2, D3 and D4) couple to Gαi/Gα0 proteins and inhibit adenylate cyclase leading to reduced cyclic AMP levels 35, 36. However, recently, dopamine D1‐like receptors have also been found to couple to increases in intracellular calcium probably through Gαq followed by the activation of phospholipase C and release of intracellular calcium by inositol triphosphate (IP3) 35, and that may explain the dopamine D1 receptor‐induced increase in calcium and nitric oxide formation (fig. 1). Also the D2‐like receptors and/or heterodimers of the dopamine receptors have been described to couple to other signal transduction pathways both through Gβγ and arrestin 35. Therefore, due to biased agonism of some of the agonists, the functional effects of agonists with apparently same binding affinity may differ considerably.
Apomorphine
There is an extensive literature on apomorphine and sexual function 1, 37. In brief, apomorphine is a general dopamine receptor agonist with reported receptor binding affinity (Ki) for dopamine receptors as follows: D1‐like [D1 (101 nM), D5 (10 nM)] and D2‐like [D2 (32 nM), D3 (26 nM), and D4 (2.6 nM)] 38 (table 1). In the supraoptic nucleus (SON), MPOA, and the PVN of the hypothalamus, oxytocinergic neurons express D2, D3 and D4 receptors, and apomorphine was suggested to induce erection through the activation of D2‐like receptors in the PVN 39. Activation of spinal dopamine D2 receptors may also play a role in the pro‐erectile effect of apomorphine as mentioned above 23. Matsumoto et al. 40 found that in isolated rat erectile tissue apomorphine‐induced relaxation appears to involve dopamine D1 and D2 receptors, but the authors concluded that ‘at relevant systemic doses of apomorphine, peripheral effects of the compound are unlikely to contribute to its pro‐erectile effects in rats’. Therefore, the binding profile and the studies of erectile function support mainly an involvement of central D2‐like receptors in the pro‐erectile effect of apomorphine.
Apomorphine was developed for the treatment of Parkinson’s disease, but a major drawback is the induction of nausea and vomiting. However, by sublingual administration of apomorphine, a tolerable therapeutic window was found allowing induction of erection and lowering of the incidence of side effects (yawning, nausea, vomiting, dizziness, blurred vision, diaphoresis, pallor, mild hypotension and bradycardia) observed by intranasal apomorphine administration 41. In a phase III study of the effect of apomorphine on ED with moderate‐to‐severe dysfunction in 74% of the men, apomorphine‐induced erections occurred 10–25 min. after administration. Moreover, apomorphine in this study did lead to erection suitable to intercourse in 54.4% versus 33.8% in the placebo group 42. However, in a post‐marketing observational cohort study in 11,185 men presenting with ED in general practice, the main reason for stopping treatment with apomorphine was that the drug was ineffective in 64.7% of the patients 43.
As mentioned, apomorphine has a series of potential adverse effects. The most frequent side effect to the sublingual formulation of apomorphine in the post‐marketing study was headache 43, and similar to hypotension and yawning, this effect is probably predictable by the effect on dopamine receptors. However, apomorphine has also been shown in low concentrations to activate transient receptor potential ankyrin 1 (TRPA1) channels involved in pain sensation, inflammation and nausea 44, suggesting that off target binding contributes to the side effects of apomorphine.
Selective dopamine D1 receptor agonists and erectile function
In the first studies of the effect of apomorphine microinjected in PVN, the pro‐erectile effect was blocked both by the dopamine D1 receptor antagonist, SCH 23390, and dopamine D2 receptor antagonists, haloperidol and (−)‐sulpiride 12. Fenoldopam is a benzazepine derivative and a selective partial agonist primarily at dopamine D1 receptors (table 1) and has no significant affinity for dopamine D2 receptors, but binds with moderate affinity to α2‐adrenoceptors. It is registered as an antihypertensive drug mainly for treating hypertensive crisis and/or peripheral vascular disease. Systemic infusion of fenoldopam dose‐dependently potentiated the increases in intracavernosal pressure (a measure of erectile function) elicited by submaximal electric stimulation of the distal part of the cavernous nerve in anaesthetized rats 45. Although in vitro studies showed an involvement of nitric oxide in relaxations of corpus cavernosum induced by fenoldopam and another dopamine D1 receptor selective agonist, A‐68930 31, 45, in vivo the pro‐erectile effect of fenoldopam persisted in the presence of an inhibitor of NOS suggesting that there is a direct pro‐erectile effect of fenoldopam. However, these findings need to be confirmed by other studies, and it would also be interesting to clarify whether local administration or systemic administration of fenoldopam in intact conscious animals and/or man leads to erection.
Selective dopamine D2 and D3 receptor agonists and erectile function
Similar to apomorphine, dopamine D2/D3 agonists, bromocriptine and quinelorane, induce spontaneous erections and increase the number of erectile events in rats 46–48, probably by the activation of oxytocinergic neurons in the PVN of rats 47. Dopamine D2 and D3 receptors have 50% homology in structure, and it has proven difficult to obtain compounds selective for dopamine D3, as opposed to D2, receptors 49. Dopamine D2 receptors have been the target for the treatment of movement disorders related to Parkinson’s disease with dopamine D2 agonists, while modulation of dopamine D3 receptors is of interest for the treatment of Parkinson’s disease, drug addiction and schizophrenia 49, 50. Several of the dopamine agonists, for example bromocriptine, cabergoline, pergolide, ropinirole and rotigotine, used for the treatment of this disease have dopamine D2 receptor agonist effects (table 1), and they can also be associated with increased libido (hypersexuality). Prolonged erection (>4 hr) defined as priapism in man has been described in case reports for cabergoline and rotigotine 51, 52 and is devastating for erectile function. In addition to effect on dopamine D2 receptors, cabergoline and rotigotine both bind to and antagonize α1‐adrenoceptors, and antagonism of α1‐adrenoceptors is a frequent cause of priapism 53.
The number of studies examining the effect of dopamine D2 and D3 agonists for the treatment of sexual dysfunction is limited. In one open trial where 13 patients reported sexual dysfunction after they began with antidepressant treatment, sexual function, measured by use of the Arizona Sexual Experience Scale and Clinical Global Impression of Improvement scales, showed a significant effect on both scales 3 weeks after initiation of treatment with ropinirole 54. The doses of ropinirole (3–4 mg/day) in the latter study were markedly lower than the doses (20–25 mg/day) described to cause hypersexuality and sexual urges in patients with Parkinson’s disease 10, 54. Therefore, the potential for treating ED of mixed origin by selective agonists for dopamine D2 or D3 receptors remains to be explored, although the effects on potentially severe side effects as behaviour and blood pressure should be taken into account. Moreover, it would be relevant to conduct randomized, double‐blind studies using low doses of these agonists for treating antidepressant‐induced sexual dysfunction.
Selective dopamine D4 receptor agonists and erectile function
A selective dopamine D4 receptor agonist, PD168077, was found to induce erection in rats when microinjected into the PVN 47, 55, 56. However, by staining for Fos in oxytocinergic neurons in the PVN, PD168077, in contrast to quinelorane, was not found to activate these neurons. Moreover, the effect of PD168077 was context dependent; for example, the effect was observed in the presence of receptive female rats and may involve dopamine D4 receptor activation of the magnocellular oxytocin system 47.
Apomorphine has high affinity for dopamine D4 receptors, but it does also bind to the dopamine D2 receptors with high affinity (table 1). To avoid the dose‐limiting side effects mediated by D2 receptors, for example nausea and emesis, dopamine D4 receptor agonists, ABT 724 and ABT 670, were developed for the treatment of ED. In freely moving rats, increases in intracavernosal pressure were measured and showed that ABT 724 administered subcutaneously dose‐dependently increased the incidence and magnitude of spontaneous erections, and these responses were larger and of longer duration in the presence of a PDE5i, sildenafil 57. Injection in ferrets, as a pre‐clinical model for nausea and emesis, revealed that in contrast to a selective dopamine D2 receptor agonist, PNU‐95666E, ABT 724 in rather high doses did not have these effects 57. Another dopamine D4 receptor agonist, ABT 670, with better oral bioavailability was developed, but it has markedly less affinity to dopamine D4 receptors compared with ABT 724 (table 1, 58). Administered subcutaneously, incidences of ABT 670‐induced penile erections were less than observed with the same dose of apomorphine 58. These data agree with the observations that the ABT 724 and ABT 670 are less potent and have less efficacy compared with apomorphine, probably also explained by the observations that they are partial agonists at dopamine D4 receptors 57, 58. The development of these dopamine D4 agonists, ABT 724 and ABT 670, reached, respectively, phase I and phase II trials. Therefore, despite apparently less adverse effects with ABT 724 and ABT 670 compared with apomorphine, potency and efficacy were lower. Here, also the finding that the effect on erectile function of dopamine D4 receptors is context dependent can play a role for the outcome.
Selective dopamine D5 receptor agonists and erectile function
Information regarding effect of dopamine D5 receptor agonists on sexual function is limited. Several studies have been performed to identify the areas of expression of dopamine D5 receptors. The dopamine D5 receptor is expressed in the hypothalamus and the parafascicular nucleus of the thalamus in the brain of the rat and human being. The different expression areas for dopamine D5 receptors versus other dopamine receptors suggest that dopamine D5 receptors play a role as regulator and integrator of dopaminergic function 59. The dopamine D5 receptors are also expressed in other brain areas, for example substantia nigra‐pars compacta, striatum, cerebral cortex, nucleus accumbens and the olfactory tubercle 60. A recent report showed that knockout of the dopamine D5 receptors diminishes the sexual receptivity in female mice and in males influences the rewarding effects of pre‐ejaculatory penile intromission 61. Reduction in sexual interest, ED and ejaculatory problems are present in patients with antipsychotics 62. Therefore, a dopamine D5 receptor agonist could be a supplement to improve sexual disposition and probably libido in these patients.
In summary, agonist stimulation of dopamine receptors is a way forward to obtain pro‐erectile effects, and both central and peripheral dopamine receptors are potential targets for the treatment of ED. It seems that the erectile effects are dependent on the activation of dopamine D2, D3, D4 receptors in CNS and in the periphery mainly dependent on dopamine D1 receptor activation in corpus cavernosum (fig. 1). Although apomorphine appeared as a promising drug for the treatment of ED, it represents the first generation. The requirement for the next generation will be that the drug candidates have at least the same efficacy at, for example, D3/D4 receptors and at the same time avoid the side effects of apomorphine.
Dopamine receptor antagonists and erectile function
Antipsychotics and erectile function
Most antipsychotic drugs bind to and antagonize the effect of dopamine at dopamine D2 receptors, for example haloperidol. Therefore, older anti‐psychotic drugs are often associated with decreased erection and anorgasmia in 30–60% of the patients, while newer anti‐psychotics appear to have lower incidence of sexual dysfunction 5, 62. Among the newer anti‐psychotics, risperidone appears to have the highest rate of sexual dysfunction, while there are insufficient data on aripiprazole and ziprasidone. A pilot study switching treatment from the atypical antipsychotics, risperidone, amisulpride and olanzapine to aripiprazole apparently improved sexual performance in a small patient population 63, but this needs to be confirmed by studies with larger numbers of patients.
It is difficult to separate disease from the drug effect and to obtain reliable information from patients with psychotic illness. Thus, the sexual dysfunction associated with the antipsychotic treatment not only affects erectile function, but also reduces libido, causes retrograde ejaculation and reduces sexual arousal and overall sexual satisfaction. A recent Cochrane review examined the possibilities for intervention against sexual dysfunction in patients in treatment with antipsychotics and suggested that switching to olanzapine may improve sexual functioning both in men and women 64. However, these results were based on a small open‐label trial, and another possibility is to treat the patients with schizophrenia and ED with sildenafil, but again the evidence is sparse 64.
Buspirone
Buspirone is an anxiolytic drug with less affection of sexual function compared with SSRIs and may even reverse the sexual dysfunction associated with SSRI treatment 65, 66. Buspirone is a partial agonist on 5‐hydroxytryptamine type 1A (5‐HT1A receptors), but both buspirone and the main 5‐and 6‐hydroxylmetabolites bind to dopamine D3 and D4 receptors with affinities comparable to the binding to 5‐HT1A receptors, and they function as antagonists at both dopamine D3 and D4 receptors 67. Activation of 5‐HT1A receptors by agonists, for example 8‐OH‐DPAT, inhibits penile erection in rats, while antagonists of this receptor enhance the acute pro‐erectile effect of subcutaneously administered apomorphine and fluoxetine 68. Therefore, the less affection of erectile function observed with buspirone versus SSRI can probably be ascribed to buspirone acting as antagonist on the 5‐HT1A receptors, but further studies will be required to fully understand the mechanism underlying the effect of buspirone versus SSRI on erectile function.
Drugs increasing the synaptic dopamine content
The synaptic dopamine content can be increased either by increased release of dopamine, by inhibition of the dopamine reuptake transporter (DAT), or by blocking dopamine breakdown by inhibition of catechol‐O‐methyltransferase (COMT) or monoamine oxidase (MAO) type B inhibitors.
Release of dopamine
Amantadine
The antiviral drug amantadine stimulates the release of dopamine from nerve terminals and inhibits the reuptake of dopamine in the presynaptic neuron. In addition, it is a weak antagonist at N‐methyl‐d‐aspartate (NMDA) receptors. Despite a long half‐life of 10–15 hr in man, the effects are thought to be short‐lived as there is development of tolerance to the drug. In several case reports where men and women were in treatment with antidepressants, for example fluoxetine and paroxetine, it has been reported that addition of treatment with amantadine reversed the sexual dysfunction (anorgasm, ED) 10, 69. However, randomized trials will be needed to evaluate whether amantadine is in fact useful for reversal of sexual dysfunction in patients treated with antidepressants.
Amphetamine and methamphetamine
Oral methamphetamine is used for the treatment of attention deficit hyperactivity disorder and also as recreational drug. Amphetamine has been reported to arouse sexual behaviour 70. Amphetamine is associated with increased release of dopamine, but also increased release of noradrenaline from sympathetic nerves which will lead to constriction of penile arteries and of corpus cavernosum 71. Therefore, methamphetamine abuse and/or chronic use often lead to ED, an effect which can also be reproduced in rats 72. PDE5i, for example tadalafil, improves erectile function in rats chronically treated with methamphetamine 72, and this may explain the recreational use combining methamphetamine/amphetamine with the intake of a PDE5i 73, 74.
Clavulanic acid
Clavulanic acid is a beta‐lactamase inhibitor and is often used in combination with penicillin to prevent the breakdown of the penicillin, for example amoxicillin, by bacterial beta‐lactamase. Clavulanic acid is orally active and stable and approximately 25% reaches the cerebrospinal fluid in man. Testing non‐human primates with clavulanic acid was found to increase sexual arousal 75, and therefore the drug was tested in male rats exposed to female rats brought into sexual behavioural oestrus by injection of 50 μg estradiol benzoate, and the number of intromissions and ejaculations was measured 76. Elimination half‐life of clavulanic acid is approximately 1 hr 77, but only minor acute pro‐sexual activity was observed at the highest dose administered. However, after 7 days of treatment, clavulanic acid increased both the number of ejaculations and intromissions, while the SSRI, paroxetine, reduced sexual activity 76. Apparently, clavulanic acid did not bind to a series of classical neurotransmitter receptors 75, but in cultured neuronal cell lines (PC12 and SH‐SY5Y cells) stimulation with a relatively high clavulanic acid concentration (100 μM) increased the release of dopamine, probably by binding to two proteins of importance for neurosecretion and vesicle transport 78. The expected cerebrospinal concentrations of clavulanic acid with anxiolytic and erectile effects are in the range of 1–10 pM 75. That this mechanism could play a role was further supported by the observations that a dopamine D2 receptor antagonist antagonizes the pro‐erectile effect of clavulanic acid in rats 79. In the latter study, the effect of clavulanic acid administered by intraperitoneal injection was compared to apomorphine, and while apomorphine induced concentration‐dependent increases in the number of spontaneous erections, the concentration–response curve for clavulanic acid was bell‐shaped with maximum effect at 1 mg/kg and then decreasing with higher doses 80. The authors suggested that in addition to incrementing dopamine release, clavulanic acid may also increase release of serotonin acting at 5‐HT2C receptors, as mianserin also antagonized the pro‐erectile effect of clavulanic acid. A phase II study was planned for examining the efficacy of clavulanic in patients, but it was suspended 11. A series of issues remain to be solved for clavulanic acid including the target(s) involved in the pro‐erectile effect and an explanation for the bell‐shaped dose–response curve for clavulanic acid on erectile function.
Inhibition of the dopamine reuptake transporter
Cocaine
Addictive drugs including cocaine are normally thought to enhance dopamine within the mesotelencephalic reward/reinforcement circuity of the forebrain, mainly in the nucleus accumbens 81, but other pathways observed by magnetic resonance image including subcallosal cortex caudate, putamen, basal forebrain, thalamus, insula, hippocampus, parahippocampal gyrus, cingulate, temporal cortices, parietal cortex, extra‐striatal cortices and pons also play an important role in cocaine abuse 82. Moreover, the pharmacokinetics and rapid penetrance to CNS play an important role for the abuse of cocaine 83. Cocaine inhibits the pre‐synaptic transporters for noradrenaline (NET), dopamine (DAT) and serotonin (SERT), which increase the synaptic concentrations of noradrenaline, dopamine and serotonin. Cocaine may increase sexual arousal and lead to penile erection 84, but chronic use of cocaine is associated with ED in man 85. D1/D5 antagonists like SCH39166 (ecopipam) can attenuate the euphoric effects of cocaine 86. Centrally administered cocaine apparently inhibits penile erection in rats via noradrenergic neurotransmission, activating α‐adrenoceptors in hippocampus 87. At peripheral level, cocaine‐induced endothelial dysfunction with increased formation of endothelin‐1 and reduced nitric oxide and vasoconstriction was found to contribute to cocaine‐induced ED 85. These findings show a good relation between normal erectile function of brain‐activated areas and cocaine brain‐activated areas, which suggests that drugs for improving erectile function with effect on the monoamine transporters should have selectivity for the dopamine transporter. Moreover, a drug without a fast onset of the inhibition of the dopamine reuptake should be aimed for to avoid abuse as observed for cocaine.
Bupropion
Antidepressants act with varying degree on the monoamine transporters. The selectivity is high for the inhibition of SERT by the SSRI, for example fluoxetine, and these drugs are often associated with sexual dysfunction. Depression by itself makes it difficult to separate effect of illness from additive effect of drugs. Mood disorders may lead to lack of interest and emotional withdrawal from the sexual partner. Antidepressants can have numerous effects on sexual function including reduced sexual desire, erection difficulties and orgasm problems. SSRI and venlafaxine can negatively affect all the steps of the male sexual response cycle (desire/arousal–excitement–orgasm). As summarized based on fifteen randomized studies, bupropion, nefazodone and mirtazapine have lower rates of sexual dysfunction than SSRIs 9, 88, and bupropion can be used to alleviate sexual symptoms caused by other antidepressant medication 89, 90. Bupropion is so far the only antidepressant with some selectivity for DAT over NET and SERT with Ki values (nM) of, respectively, 526, 52,600 and 9100 for the three transporters. Infusion of bupropion in the MPOA in rats increased dopamine measured by microdialysis and evoked penile erections in rats 91, but a rather high concentration (10 mM) of bupropion had to be applied to observe the effect 91 suggesting either that the MPOA is not the primary location where bupropion acts or that the drug during acute infusion activates other anti‐erectile pathways. Indeed, acute systemic administration of bupropion reduced penile erections in rats, while bupropion treatment for 14 days (chronic treatment) was not associated with changes in penile erections in rats, which was in contrast to treatment with fluoxetine or desipramine, an inhibitor of primarily NET 92. These findings suggest that not only an up‐regulation of dopamine in the synaptic cleft is probably sufficient to explain a pro‐erectile effect. Changes in dopamine content are associated with altered dopamine receptor expression 35, 93 and may explain differences in the acute and chronic effects on erectile function of centrally acting inhibitors of monoamine transporters.
Breakdown of dopamine
Selegiline
Selegiline is a selective, irreversible inhibitor of MAO B and used alone or in combination with levodopa in the treatment of Parkinson’s disease. The inhibition of MAO B will delay the degradation and prolong the action of dopamine. Selegiline undergoes metabolism to amphetamine and methamphetamine, which may also contribute to the pharmacological effects on erectile function as described above for amphetamine and methamphetamine. Selegiline enhances the effect of dopamine in the PVN in rats 94. Therefore, selegiline or vehicle was applied for 3 weeks for the treatment of sexual dysfunction in patients in antipsychotic treatment in placebo‐controlled, crossover study. Apart from reduction in prolactin induced by selegiline, there was no difference in sexual function compared with the placebo treatment period 95.
Catechol‐O‐methyltransferase inhibitors
The COMT inhibitors entacapone and tolcapone are used as adjunct to levodopa for end‐of‐dose deterioration in Parkinson’s disease and they increase levodopa availability in the brain. Only tolcapone penetrates to the brain and increases dopamine in pre‐frontal cortex and ventral hippocampus, but this effect is more pronounced in female compared with male rats 96. However, it is unclear whether inhibition of peripheral COMT also increasing noradrenaline will counteract the central effect of increasing dopamine on sexual function, and this remains to be clarified.
Modulation of dopaminergic pathways and ejaculation
When considering drugs for the treatment of ED, the effect on ejaculation is also highly relevant. The physiology and pharmacology of ejaculation has recently been extensively reviewed 97, 98, and here is only a brief description of the role of the dopaminergic pathways in ejaculation. The incertohypothalamic pathway that includes MPOA seems to be involved in the control of the ejaculatory response. In either case, increases in extracellular dopamine levels in MPOA and injection of apomorphine in the MPOA induce shortening of the ejaculation latency with increased number of ejaculations and copulatory rate in male rats 97. The dopamine D3 receptor seems to play an important role, as the D2/D3 receptor agonist 7‐hydroxy‐N,N‐di‐n‐propyl‐2‐aminotetralin (7‐OH‐DPAT) enhances ejaculatory behaviour, while blockade of dopamine D3 receptors with highly selective antagonists prolongs ejaculation latency and the post‐ejaculatory refractory period 99. Therefore, D3 receptor antagonists have also been proposed for the treatment of premature ejaculation 97, although 7‐OH‐DPAT had shown a partial recovery in ejaculatory reflexes in a model of spinal cord contusion injury 100. Polymorphism with increased numbers of repeats of the DAT1 transporter gene has been suggested to be linked to human premature ejaculation 101, while it is less clear whether an inhibitor of DAT, bupropion, in therapeutic concentrations affects ejaculation. Dapoxetine and off‐label SSRIs are efficacious and safe for the treatment of premature ejaculation, while it remains to be investigated in man whether there would be a beneficial effect of dopamine D3 receptor antagonists. The pathways involved in ejaculation are still incompletely understood 98, and considering drugs altering specifically the dopaminergic pathways for the treatment of ED, it would be relevant to examine also the effect on ejaculation in health and disease to ensure overall sexual function.
Modulation of Dopaminergic Pathways and Unmet Needs in Patients with Erectile Dysfunction
Despite PDE5 inhibitors are efficacious drugs, there are still large populations of patients where this treatment is insufficient or can be optimized including patients with ED suffering from diabetes, metabolic syndrome, neurodegenerative disease and patients who have undergone radical prostatectomy affecting the cavernous nerves. In the following, we have focused on diabetes and neurodegenerative disease where we find drugs modulating the dopaminergic pathways will be of interest. Many of the considerations regarding metabolic syndrome are similar to those of diabetes, while we find nerve lesions in connection with radical prostatectomy or pelvic surgery require other approaches and this has been addressed elsewhere 102.
Diabetes and erectile function
Diabetes is associated with a markedly higher risk of ED than in the general population 103–105. In clinics that treat ED, the prevalence of diabetes as comorbidity is 20–25% 106. Intensive glycaemic control reduces the prevalence of ED 10 years later in young men with type 1 diabetes 107. However, there is lack of efficacy of PDE5 inhibitors in 50% of the patients suffering from diabetes and ED 8, 108–110. These observations suggest that in addition to affection of the peripheral tissue involved in erection, diabetes also affects the complex interaction of the sensory and autonomic nerves involved in erection.
The affection of the erectile tissue and peripheral parasympathetic nerves in diabetes is well described. In brief, endothelial cell dysfunction, smooth muscle dysfunction and autonomic neuropathy have been described in diabetic rats and in isolated tissue from diabetic men 111, 112 (see Ref. 5). Moreover, hypogonadism is more prevalent in patients with diabetes and ED compared with non‐diabetics with ED. Due to lack of efficacy of PDE5 inhibitors, supplementation with testosterone and also drugs targeting the Rho kinase pathway has been investigated and tested in patients with ED and diabetes 113–115.
The affection by diabetes of central pathways involved in erectile function is less well described. However, the prevalence of ED in patients with diabetic neuropathy is close to 100% 116. In rats with spontaneous development of diabetes, the BB/WOR rat, both central and peripheral neuropathies, was found involved in the sexual dysfunction 117. Reduction in microvascular flow has been suggested to play a role for the development of peripheral neuropathy associated with ED 118. Other mechanims seem also to play a role. In streptozotocin‐induced type 1 diabetic rats, NMDA‐induced erection was investigated by direct injection in the PVN 119. NMDA stimulates the same oxytocinergic pathways as dopamine, and there was ED and reduced expression of neuronal NOS in PVN 119. The authors found that both exercise and treatment with angiotensin II type 1 (AT1) receptor antagonists improve erectile function in the streptozocin‐induced diabetic rat model 120, 121, but the effect of dopaminergic drugs on erectile function remains to be investigated in animal models with diabetes.
There is evidence supporting that diabetes affects the central dopaminergic pathways and leads to disease. The risk of Parkinson’s disease was ~40% higher among patients with diabetes than among patients without diabetes 122, and insulin resistance in the brain of brain‐specific knockout of the insulin receptor induces increased levels of monoaminoxidase type B, increased dopamine turnover and decreased dopamine release 123. In mice with high fat‐induced diabetes, there is a loss of dopaminergic neurons 124. These findings suggest that affection of dopaminergic pathways in diabetes may also play a role in ED. However, the centrally acting dopaminergic agonist, apomorphine, induced only effect comparable to placebo in 130 patients with diabetes and ED, and the typical responder was young and with good glycaemic control 125. Further studies will be required to understand whether modulation of the dopaminergic pathways not only at central, but also at peripheral level may restore erectile function in diabetes.
Neurodegenerative disease and erectile dysfunction
The effects of the neurodegenerative diseases, Parkinson’s disease and Alzheimer’s disease, on sexual dysfunction and recommended treatment have recently been reviewed by others 126. Sexual dysfunction is common in patients with these disorders, and ED is considered one of the most distressing associated disabilities 127. PDE5i and apomorphine are efficacious in most patients with Parkinson’s disease and ED 42, although higher doses than the recommended of apomorphine will be required 127. As mentioned in relation to the different dopaminergic drugs in the sections above, drug‐induced hypersexuality also has to be dealt with to provide an optimal treatment with dopaminergic drugs in Parkinson’s disease.
Conclusions and Perspectives
We have focused on the modulation of the dopaminergic pathway for the treatment of ED. A series of dopaminergic agonists have effect on erectile function in rodents with central activation of dopamine D2‐like receptors and mainly of D1‐like receptors in the erectile tissue. The therapeutic window for the general dopamine agonist apomorphine is narrow and it is mainly acting on central dopamine receptors. Therefore, apomorphine was found to fail in patients with ED and diabetes and to be less efficacious compared with PDE5 inhibitors. The present MiniReview of the literature proposes that modulation of the dopaminergic pathways provides a possibility to improve the treatment of ED. However, this may require a novel approach targeting the dopaminergic pathway at several levels of the penile reflex arch. Moreover, further testing of dopaminergic drugs in animal models for diabetes, metabolic syndrome and/or neurodegenerative disease would be relevant.
Acknowledgements
Ulf Simonsen is part of MEMBRANES, Aarhus University, while Simon Comerma‐Steffensen was supported by a PhD grant from Aarhus University.
Conflict of Interest
The authors declare no conflict of interest.
- 1Andersson KE. Mechanisms of penile erection and basis for pharmacological treatment of erectile dysfunction. Pharmacol Rev 2011;63:811–59.
- 2Melis MR, Argiolas A. Central control of penile erection: a re‐visitation of the role of oxytocin and its interaction with dopamine and glutamic acid in male rats. Neurosci Biobehav Rev 2011;35:939–55.
- 3Giuliano F, Bernabe J, Jardin A, Rousseau JP. Antierectile role of the sympathetic nervous system in rats. J Urol 1993;150:519–24.
- 4Dail WG, Barba V, Leyba L, Galindo R. Neural and endothelial nitric oxide synthase activity in rat penile erectile tissue. Cell Tissue Res 1995;282:109–16.
- 5Gratzke C, Angulo J, Chitaley K, Dai YT, Kim NN, Paick JS et al. Anatomy, physiology, and pathophysiology of erectile dysfunction. J Sex Med 2010;7:445–75.
- 6Goldstein I, Jones LA, Belkoff LH, Karlin GS, Bowden CH, Peterson CA et al. Avanafil for the treatment of erectile dysfunction: a multicenter, randomized, double‐blind study in men with diabetes mellitus. Mayo Clin Proc 2012;87:843–52.
- 7Porst H, Burnett A, Brock G, Ghanem H, Giuliano F, Glina S et al. SOP conservative (medical and mechanical) treatment of erectile dysfunction. J Sex Med 2013;10:130–71.
- 8Nehra A, Jackson G, Miner M, Billups KL, Burnett AL, Buvat J et al. The Princeton III Consensus recommendations for the management of erectile dysfunction and cardiovascular disease. Mayo Clin Proc 2012;87:766–78.
- 9Taylor MJ, Rudkin L, Hawton K. Strategies for managing antidepressant‐induced sexual dysfunction: systematic review of randomised controlled trials. J Affect Disord 2005;88:241–54.
- 10Moll JL, Brown CS. The use of monoamine pharmacological agents in the treatment of sexual dysfunction: evidence in the literature. J Sex Med 2011;8:956–70.
- 11Peak TC, Yafi FA, Sangkum P, Hellstrom WJ. Emerging drugs for the treatment of erectile dysfunction. Expert Opin Emerg Drugs 2015;20:263–75.
- 12Melis MR, Argiolas A, Gessa GL. Apomorphine‐induced penile erection and yawning: site of action in brain. Brain Res 1987;415:98–104.
- 13Argiolas A, Hedlund H. The pharmacology and clinical pharmacokinetics of apomorphine SL. BJU Int 2001;88(Suppl 3):18–21.
- 14Argiolas A, Melis MR, Mauri A, Gessa GL. Paraventricular nucleus lesion prevents yawning and penile erection induced by apomorphine and oxytocin but not by ACTH in rats. Brain Res 1987;421:349–52.
- 15Chen KK, Chan JY, Chang LS. Dopaminergic neurotransmission at the paraventricular nucleus of hypothalamus in central regulation of penile erection in the rat. J Urol 1999;162:237–42.
- 16Hull EM, Muschamp JW, Sato S. Dopamine and serotonin: influences on male sexual behavior. Physiol Behav 2004;83:291–307.
- 17Bjorklund A, Lindvall O, Nobin A. Evidence of an incerto‐hypothalamic dopamine neurone system in the rat. Brain Res 1975;89:29–42.
- 18Skagerberg G, Lindvall O. Organization of diencephalic dopamine neurones projecting to the spinal cord in the rat. Brain Res 1985;342:340–51.
- 19Skagerberg G, Bjorklund A, Lindvall O, Schmidt RH. Origin and termination of the diencephalo‐spinal dopamine system in the rat. Brain Res Bull 1982;9:237–44.
- 20Ridet JL, Sandillon F, Rajaofetra N, Geffard M, Privat A. Spinal dopaminergic system of the rat: light and electron microscopic study using an antiserum against dopamine, with particular emphasis on synaptic incidence. Brain Res 1992;598:233–41.
- 21Holstege JC, Van DH, Buijs RM, Goedknegt H, Gosens T, Bongers CM. Distribution of dopamine immunoreactivity in the rat, cat and monkey spinal cord. J Comp Neurol 1996;376:631–52.
- 22Van DH, Dijk J, Voom P, Holstege JC. Localization of dopamine D2 receptor in rat spinal cord identified with immunocytochemistry and in situ hybridization. Eur J Neurosci 1996;8:621–8.
- 23Giuliano F, Allard J. Dopamine and male sexual function. Eur Urol 2001;40:601–8.
- 24Giuliano F, Allard J, Rampin O, Droupy S, Benoit G, Alexandre L et al. Spinal proerectile effect of apomorphine in the anesthetized rat. Int J Impot Res 2001;13:110–5.
- 25Hurt KJ, Musicki B, Palese MA, Crone JK, Becker RE, Moriarity JL et al. Akt‐dependent phosphorylation of endothelial nitric‐oxide synthase mediates penile erection. Proc Natl Acad Sci USA 2002;99:4061–6.
- 26Simonsen U, Garcia‐Sacristan A, Prieto D. Penile arteries and erection. J Vasc Res 2002;39:283–303.
- 27Schjorring O, Kun A, Flyvbjerg A, Kirkeby HJ, Jensen JB, Simonsen U. Flow‐evoked vasodilation is blunted in penile arteries from Zucker diabetic fatty rats. J Sex Med 2012;9:1789–800.
- 28Lue TF, Tanagho EA. Physiology of erection and pharmacological management of impotence. J Urol 1987;137:829–36.
- 29El‐Din MM, Senbel AM, Daabees TT, Sharabi FM. Peripheral modulation of dopaminergic receptors affects erectile responses in rats. Basic Clin Pharmacol Toxicol 2007;100:225–32.
- 30Hyun JS, Bivalacqua TJ, Baig MR, Yang DY, Leungwattanakij S, Abdel‐Mageed A et al. Localization of peripheral dopamine D1 and D2 receptors in rat corpus cavernosum. BJU Int 2002;90:105–12.
- 31d’Emmanuele di Villa Bianca R, Sorrentino R, Roviezzo F, Imbimbo C, Palmieri A, De Dominicis G et al. Peripheral relaxant activity of apomorphine and of a D1 selective receptor agonist on human corpus cavernosum strips. Int J Impot Res 2005;17:127–33.
- 32Han DH, Chae MR, So I, Park JK, Lee SW. The effects of dopamine receptor agonists on BK Ca channels and signal transduction mechanism in corpus cavernosal smooth muscle cells. Int J Impot Res 2008;20:53–9.
- 33Mignini F, Traini E, Tomassoni D, Amenta F. Dopamine plasma membrane transporter (DAT) in rat thymus and spleen: an immunochemical and immunohistochemical study. Auton Autacoid Pharmacol 2006;26:183–9.
- 34Pfeil U, Kuncova J, Bruggmann D, Paddenberg R, Rafiq A, Henrich M et al. Intrinsic vascular dopamine – a key modulator of hypoxia‐induced vasodilatation in splanchnic vessels. J Physiol 2014;592:1745–56.
- 35Beaulieu JM, Espinoza S, Gainetdinov RR. Dopamine receptors – IUPHAR Review 13. Br J Pharmacol 2015;172:1–23.
- 36Kebabian JW. Multiple classes of dopamine receptors in mammalian central nervous system: the involvement of dopamine‐sensitive adenylyl cyclase. Life Sci 1978;23:479–83.
- 37Heaton JP. Key issues from the clinical trials of apomorphine SL. World J Urol 2001;19:25–31.
- 38Hsieh GC, Hollingsworth PR, Martino B, Chang R, Terranova MA, O’Neill AB et al. Central mechanisms regulating penile erection in conscious rats: the dopaminergic systems related to the proerectile effect of apomorphine. J Pharmacol Exp Ther 2004;308:330–8.
- 39Baskerville TA, Douglas AJ. Interactions between dopamine and oxytocin in the control of sexual behaviour. Prog Brain Res 2008;170:277–90.
- 40Matsumoto K, Yoshida M, Andersson KE, Hedlund P. Effects in vitro and in vivo by apomorphine in the rat corpus cavernosum. Br J Pharmacol 2005;146:259–67.
- 41Heaton JP, Morales A, Adams MA, Johnston B, el Rashidy R. Recovery of erectile function by the oral administration of apomorphine. Urology 1995;45:200–6.
- 42Heaton JP, Altwein JE. The role of apomorphine SL in the treatment of male erectile dysfunction. BJU Int 2001;88(Suppl 3):36–8.
- 43Maclennan KM, Boshier A, Wilton LV, Shakir SA. Examination of the safety and use of apomorphine prescribed in general practice in England as a treatment for erectile dysfunction. BJU Int 2006;98:125–31.
- 44Schulze A, Oehler B, Urban N, Schaefer M, Hill K. Apomorphine is a bimodal modulator of TRPA1 channels. Mol Pharmacol 2013;83:542–51.
- 45Senbel AM. Interaction between nitric oxide and dopaminergic transmission in the peripheral control of penile erection. Fundam Clin Pharmacol 2011;25:63–71.
- 46Sharifzadeh M, Dehpour AR, Samini M, Hassan‐Mazandarani H, Samadian T, Asghari GR. Alterations of bromocriptine‐induced penile erection by chronic lithium in rats. J Psychopharmacol 1996;10:157–61.
- 47Baskerville TA, Allard J, Wayman C, Douglas AJ. Dopamine–oxytocin interactions in penile erection. Eur J Neurosci 2009;30:2151–64.
- 48Radosavljevic M, Pajovic B, Radunovic M, Radojevic N, Bjelogrlic B. Influence of dihydroergotoxine, bromocriptine, and ergotamine on penile erection in Wistar rats. J Androl 2012;33:866–71.
- 49Le FB, Collo G, Rabiner EA, Boileau I, Merlo PE, Sokoloff P. Dopamine D3 receptor ligands for drug addiction treatment: update on recent findings. Prog Brain Res 2014;211:255–75.
- 50Sokoloff P, Diaz J, Le FB, Guillin O, Leriche L, Bezard E et al. The dopamine D3 receptor: a therapeutic target for the treatment of neuropsychiatric disorders. CNS Neurol Disord Drug Targets 2006;5:25–43.
- 51De La Peña Zarzuelo E, Hernández Cañas V, Llorente AC. [Priapism secondary to treatment due to cabergoline: the first description of this association]. Actas Urol Esp 2010;34:487–8.
- 52Cannas A, Meloni M, Mascia MM, Solla P, Orofino G, Farris R et al. Priapism and hypersexuality associated with rotigotine in an elderly parkinsonian patient: a case report. Clin Neuropharmacol 2016;39:162–3.
- 53Spagnul SJ, Cabral PH, Verndl DO, Glina S. Adrenergic alpha‐blockers: an infrequent and overlooked cause of priapism. Int J Impot Res 2011;23:95–8.
- 54Worthington JJ III, Simon NM, Korbly NB, Perlis RH, Pollack MH. Ropinirole for antidepressant‐induced sexual dysfunction. Int Clin Psychopharmacol 2002;17:307–10.
- 55Melis MR, Succu S, Sanna F, Melis T, Mascia MS, Enguehard‐Gueiffier C et al. PIP3EA and PD‐168077, two selective dopamine D4 receptor agonists, induce penile erection in male rats: site and mechanism of action in the brain. Eur J Neurosci 2006;24:2021–30.
- 56Melis MR, Succu S, Mascia MS, Argiolas A. PD‐168077, a selective dopamine D4 receptor agonist, induces penile erection when injected into the paraventricular nucleus of male rats. Neurosci Lett 2005;379:59–62.
- 57Brioni JD, Moreland RB, Cowart M, Hsieh GC, Stewart AO, Hedlund P et al. Activation of dopamine D4 receptors by ABT‐724 induces penile erection in rats. Proc Natl Acad Sci USA 2004;101:6758–63.
- 58Patel MV, Kolasa T, Mortell K, Matulenko MA, Hakeem AA, Rohde JJ et al. Discovery of 3‐methyl‐N‐(1‐oxy‐3′,4′,5′,6′‐tetrahydro‐2′H‐[2,4′‐bipyridine]‐1′‐ylmethyl)benzam ide (ABT‐670), an orally bioavailable dopamine D4 agonist for the treatment of erectile dysfunction. J Med Chem 2006;49:7450–65.
- 59Meador‐Woodruff JH, Mansour A, Grandy DK, Damask SP, Civelli O, Watson SJ Jr. Distribution of D5 dopamine receptor mRNA in rat brain. Neurosci Lett 1992;145:209–12.
- 60Khan ZU, Gutierrez A, Martin R, Penafiel A, Rivera A, de la Calle A. Dopamine D5 receptors of rat and human brain. Neuroscience 2000;100:689–99.
- 61Kudwa AE, Dominguez‐Salazar E, Cabrera DM, Sibley DR, Rissman EF. Dopamine D5 receptor modulates male and female sexual behavior in mice. Psychopharmacology 2005;180:206–14.
- 62Young SL, Taylor M, Lawrie SM. “First do no harm”. A systematic review of the prevalence and management of antipsychotic adverse effects. J Psychopharmacol 2015;29:353–62.
- 63Jeong HG, Lee MS, Lee HY, Ko YH, Han C, Joe SH. Changes in sexual function and gonadal axis hormones after switching to aripiprazole in male schizophrenia patients: a prospective pilot study. Int Clin Psychopharmacol 2012;27:177–83.
- 64Schmidt HM, Hagen M, Kriston L, Soares‐Weiser K, Maayan N, Berner MM. Management of sexual dysfunction due to antipsychotic drug therapy. Cochrane Database Syst Rev 2012;11:CD003546.
- 65Landen M, Eriksson E, Agren H, Fahlen T. Effect of buspirone on sexual dysfunction in depressed patients treated with selective serotonin reuptake inhibitors. J Clin Psychopharmacol 1999;19:268–71.
- 66Landen M, Bjorling G, Agren H, Fahlen T. A randomized, double‐blind, placebo‐controlled trial of buspirone in combination with an SSRI in patients with treatment‐refractory depression. J Clin Psychiatry 1998;59:664–8.
- 67Bergman J, Roof RA, Furman CA, Conroy JL, Mello NK, Sibley DR et al. Modification of cocaine self‐administration by buspirone (buspar(R)): potential involvement of D3 and D4 dopamine receptors. Int J Neuropsychopharmacol 2013;16:445–58.
- 68Protais P, Windsor M, Mocaer E, Comoy E. Post‐synaptic 5‐HT1A receptor involvement in yawning and penile erections induced by apomorphine, physostigmine and mCPP in rats. Psychopharmacology 1995;120:376–83.
- 69Balogh S, Hendricks SE, Kang J. Treatment of fluoxetine‐induced anorgasmia with amantadine. J Clin Psychiatry 1992;53:212–3.
- 70Angrist B, Gershon S. Clinical effects of amphetamine and L‐DOPA on sexuality and aggression. Compr Psychiatry 1976;17:715–22.
- 71Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI et al. Amphetamine‐type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 2001;39:32–41.
- 72Tar MT, Martinez LR, Nosanchuk JD, Davies KP. The effect of methamphetamine on an animal model of erectile function. Andrology 2014;2:531–6.
- 73Mansergh G, Shouse RL, Marks G, Guzman R, Rader M, Buchbinder S et al. Methamphetamine and sildenafil (Viagra) use are linked to unprotected receptive and insertive anal sex, respectively, in a sample of men who have sex with men. Sex Transm Infect 2006;82:131–4.
- 74Prestage G, Jin F, Kippax S, Zablotska I, Imrie J, Grulich A. Use of illicit drugs and erectile dysfunction medications and subsequent HIV infection among gay men in Sydney, Australia. J Sex Med 2009;6:2311–20.
- 75Kim DJ, King JA, Zuccarelli L, Ferris CF, Koppel GA, Snowdon CT et al. Clavulanic acid: a competitive inhibitor of beta‐lactamases with novel anxiolytic‐like activity and minimal side effects. Pharmacol Biochem Behav 2009;93:112–20.
- 76Chan JS, Kim DJ, Ahn CH, Oosting RS, Olivier B. Clavulanic acid stimulates sexual behaviour in male rats. Eur J Pharmacol 2009;609:69–73.
- 77Nakagawa H, Yamada M, Tokiyoshi K, Miyawaki Y, Kanayama T. [Penetration of potassium clavulanate/ticarcillin sodium into cerebrospinal fluid in neurosurgical patients]. Jpn J Antibiot 1994;47:93–101.
- 78Kost GC, Selvaraj S, Lee YB, Kim DJ, Ahn CH, Singh BB. Clavulanic acid increases dopamine release in neuronal cells through a mechanism involving enhanced vesicle trafficking. Neurosci Lett 2011;504:170–5.
- 79Sanna F, Succu S, Hubner H, Gmeiner P, Argiolas A, Melis MR. Dopamine D2‐like receptor agonists induce penile erection in male rats: differential role of D2, D3 and D4 receptors in the paraventricular nucleus of the hypothalamus. Behav Brain Res 2011;225:169–76.
- 80Sanna F, Melis MR, Angioni L, Argiolas A. Clavulanic acid induces penile erection and yawning in male rats: comparison with apomorphine. Pharmacol Biochem Behav 2013;103:750–5.
- 81Robinson TE, Berridge KC. The neural basis of drug craving: an incentive‐sensitization theory of addiction. Brain Res Brain Res Rev 1993;18:247–91.
- 82Breiter HC, Gollub RL, Weisskoff RM, Kennedy DN, Makris N, Berke JD et al. Acute effects of cocaine on human brain activity and emotion. Neuron 1997;19:591–611.
- 83Chow HS, Chen Z, Matsuura GT. Direct transport of cocaine from the nasal cavity to the brain following intranasal cocaine administration in rats. J Pharm Sci 1999;88:754–8.
- 84Cregler LL, Mark H. Medical complications of cocaine abuse. N Engl J Med 1986;315:1495–500.
- 85Kendirci M, Pradhan L, Trost L, Gur S, Chandra S, Agrawal KC et al. Peripheral mechanisms of erectile dysfunction in a rat model of chronic cocaine use. Eur Urol 2007;52:555–63.
- 86Romach MK, Glue P, Kampman K, Kaplan HL, Somer GR, Poole S et al. Attenuation of the euphoric effects of cocaine by the dopamine D1/D5 antagonist ecopipam (SCH 39166). Arch Gen Psychiatry 1999;56:1101–6.
- 87Chang AY, Chan JY, Chan SH. Hippocampal noradrenergic neurotransmission in concurrent EEG desynchronization and inhibition of penile erection induced by cocaine in the rat. Br J Pharmacol 2000;130:1553–60.
- 88Coleman CC, King BR, Bolden‐Watson C, Book MJ, Segraves RT, Richard N et al. A placebo‐controlled comparison of the effects on sexual functioning of bupropion sustained release and fluoxetine. Clin Ther 2001;23:1040–58.
- 89Gitlin MJ, Suri R, Altshuler L, Zuckerbrow‐Miller J, Fairbanks L. Bupropion‐sustained release as a treatment for SSRI‐induced sexual side effects. J Sex Marital Ther 2002;28:131–8.
- 90Taylor MJ, Rudkin L, Bullemor‐Day P, Lubin J, Chukwujekwu C, Hawton K. Strategies for managing sexual dysfunction induced by antidepressant medication. Cochrane Database Syst Rev 2013;5:CD003382.
- 91Adachi H, Sato Y, Kato R, Hisasue S, Suzuki K, Masumori N et al. Direct evidence of facilitative actions of dopamine in the medial preoptic area on reflexive and noncontact erections in male rats. J Urol 2003;169:386–9.
- 92Sukoff Rizzo SJ, Schechter LE, Rosenzweig‐Lipson S. A novel approach for predicting antidepressant‐induced sexual dysfunction in rats. Psychopharmacology 2008;195:459–67.
- 93Beaulieu JM, Gainetdinov RR. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol Rev 2011;63:182–217.
- 94Allard J, Bernabe J, Derdinger F, Alexandre L, McKenna K, Giuliano F. Selegiline enhances erectile activity induced by dopamine injection in the paraventricular nucleus of the hypothalamus in anesthetized rats. Int J Impot Res 2002;14:518–22.
- 95Kodesh A, Weizman A, Aizenberg D, Hermesh H, Gelkopf M, Zemishlany Z. Selegiline in the treatment of sexual dysfunction in schizophrenic patients maintained on neuroleptics: a pilot study. Clin Neuropharmacol 2003;26:193–5.
- 96Laatikainen LM, Sharp T, Harrison PJ, Tunbridge EM. Sexually dimorphic effects of catechol‐O‐methyltransferase (COMT) inhibition on dopamine metabolism in multiple brain regions. PLoS ONE 2013;8:e61839.
- 97Giuliano F, Clement P. Pharmacology for the treatment of premature ejaculation. Pharmacol Rev 2012;64:621–44.
- 98Clement P, Giuliano F. Physiology and pharmacology of ejaculation. Basic Clin Pharmacol Toxicol 2015; doi: 10.1111/bcpt.12546.
- 99Clement P, Pozzato C, Heidbreder C, Alexandre L, Giuliano F, Melotto S. Delay of ejaculation induced by SB‐277011, a selective dopamine D3 receptor antagonist, in the rat. J Sex Med 2009;6:980–8.
- 100Kozyrev N, Staudt MD, Brown A, Coolen LM. Chronic contusion spinal cord injury impairs ejaculatory reflexes in male rats: partial recovery by systemic infusions of dopamine D3 receptor agonist 7OHDPAT. J Neurotrauma 2016;33:943–53.
- 101Safarinejad MR. Relationship between premature ejaculation and genetic polymorphisms of the dopamine transporter gene (SLC6A3). BJU Int 2011;108:292–6.
- 102El‐Sakka AI. What is the current role of intracavernosal injection in management of erectile dysfunction? Int J Impot Res 2016;28:88–95.
- 103Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB. Impotence and its medical and psychosocial correlates: results of the Massachusetts Male Aging Study. J Urol 1994;151:54–61.
- 104Johannes CB, Araujo AB, Feldman HA, Derby CA, Kleinman KP, McKinlay JB. Incidence of erectile dysfunction in men 40 to 69 years old: longitudinal results from the Massachusetts male aging study. J Urol 2000;163:460–3.
- 105Martin‐Morales A, Sanchez‐Cruz JJ, Saenz de Tejada I, Rodriguez‐Vela L, Jimenez‐Cruz JF, Burgos‐Rodriguez R. Prevalence and independent risk factors for erectile dysfunction in Spain: results of the Epidemiologia de la Disfuncion Erectil Masculina Study. J Urol 2001;166:569–74.
- 106Sairam K, Kulinskaya E, Boustead GB, Hanbury DC, McNicholas TA. Prevalence of undiagnosed diabetes mellitus in male erectile dysfunction. BJU Int 2001;88:68–71.
- 107Wessells H, Penson DF, Cleary P, Rutledge BN, Lachin JM, McVary KT et al. Effect of intensive glycemic therapy on erectile function in men with type 1 diabetes. J Urol 2011;185:1828–34.
- 108Behrend L, Vibe‐Petersen J, Perrild H. Sildenafil in the treatment of erectile dysfunction in men with diabetes: demand, efficacy and patient satisfaction. Int J Impot Res 2005;17:264–9.
- 109Israilov S, Shmuely J, Niv E, Engelstein D, Livne P, Boniel J. Evaluation of a progressive treatment program for erectile dysfunction in patients with diabetes mellitus. Int J Impot Res 2005;17:431–6.
- 110Porst H, Gacci M, Buttner H, Henneges C, Boess F. Tadalafil once daily in men with erectile dysfunction: an integrated analysis of data obtained from 1913 patients from six randomized, double‐blind, placebo‐controlled, clinical studies. Eur Urol 2014;65:455–64.
- 111Saenz de Tejada I, Goldstein I, Azadzoi K, Krane RJ, Cohen RA. Impaired neurogenic and endothelium‐mediated relaxation of penile smooth muscle from diabetic men with impotence. N Engl J Med 1989;320:1025–30.
- 112Xie D, Odronic SI, Wu F, Pippen A, Donatucci CF, Annex BH. Mouse model of erectile dysfunction due to diet‐induced diabetes mellitus. Urology 2007;70:196–201.
- 113Sopko NA, Hannan JL, Bivalacqua TJ. Understanding and targeting the Rho kinase pathway in erectile dysfunction. Nat Rev Urol 2014;11:622–8.
- 114Corona G, Mannucci E, Petrone L, Ricca V, Balercia G, Mansani R et al. Association of hypogonadism and type II diabetes in men attending an outpatient erectile dysfunction clinic. Int J Impot Res 2006;18:190–7.
- 115Gur S, Peak TC, Kadowitz PJ, Sikka SC, Hellstrom WJ. Review of erectile dysfunction in diabetic animal models. Curr Diabetes Rev 2014;10:61–73.
- 116Matfin G, Jawa A, Fonseca VA. Erectile dysfunction: interrelationship with the metabolic syndrome. Curr Diab Rep 2005;5:64–9.
- 117McVary KT, Rathnau CH, McKenna KE. Sexual dysfunction in the diabetic BB/WOR rat: a role of central neuropathy. Am J Physiol 1997;272:R259–67.
- 118Cellek S, Cameron NE, Cotter MA, Muneer A. Pathophysiology of diabetic erectile dysfunction: potential contribution of vasa nervorum and advanced glycation endproducts. Int J Impot Res 2013;25:1–6.
- 119Zheng H, Bidasee KR, Mayhan WG, Patel KP. Lack of central nitric oxide triggers erectile dysfunction in diabetes. Am J Physiol Regul Integr Comp Physiol 2007;292:R1158–64.
- 120Zheng H, Liu X, Patel KP. Centrally mediated erectile dysfunction in rats with type 1 diabetes: role of angiotensin II and superoxide. J Sex Med 2013;10:2165–76.
- 121Zheng H, Mayhan WG, Patel KP. Exercise training improves the defective centrally mediated erectile responses in rats with type I diabetes. J Sex Med 2011;8:3086–97.
- 122Xu Q, Park Y, Huang X, Hollenbeck A, Blair A, Schatzkin A et al. Diabetes and risk of Parkinson’s disease. Diabetes Care 2011;34:910–5.
- 123Kleinridders A, Cai W, Cappellucci L, Ghazarian A, Collins WR, Vienberg SG et al. Insulin resistance in brain alters dopamine turnover and causes behavioral disorders. Proc Natl Acad Sci USA 2015;112:3463–8.
- 124Khang R, Park C, Shin JH. Dysregulation of parkin in the substantia nigra of db/db and high‐fat diet mice. Neuroscience 2015;294:182–92.
- 125Gontero P, D’Antonio R, Pretti G, Fontana F, Panella M, Kocjancic E et al. Clinical efficacy of apomorphine SL in erectile dysfunction of diabetic men. Int J Impot Res 2005;17:80–5.
- 126Bronner G, Aharon‐Peretz J, Hassin‐Baer S. Sexuality in patients with Parkinson’s disease, Alzheimer’s disease, and other dementias. Handb Clin Neurol 2015;130:297–323.
- 127Bronner G, Vodusek DB. Management of sexual dysfunction in Parkinson’s disease. Ther Adv Neurol Disord 2011;4:375–83.