Front. Psychiatry, 03 April 2018 | https://doi.org/10.3389/fpsyt.2018.00099
 Shuai Wang1,2,
Shuai Wang1,2,  Jing Liu3,
Jing Liu3, 
 Limin Chen1,
Limin Chen1,  Jun Wang1,
Jun Wang1,  Qunfeng Tang5,
Qunfeng Tang5,  Fuquan Zhang1 and
Fuquan Zhang1 and  Zhenhe Zhou1,2*
Zhenhe Zhou1,2*
- 1Department of Psychiatry, The Affiliated Wuxi Mental Health Center of Nanjing Medical University, Wuxi, China
- 2Wuxi Tongren International Rehabilitation Hospital, Wuxi, China
- 3Wannan Medical College, Wuhu, China
- 4Department of Substance Dependency, The Affiliated Wuxi Mental Health Center of Nanjing Medical University, Wuxi, China
- 5Department of Medical Imaging, Wuxi People’s Hospital Affiliated to Nanjing Medical University, Wuxi, China
With the rising increase in Internet-usage, Internet gaming disorder (IGD) has gained massive attention worldwide. However, detailed cerebral morphological changes remain unclear in youths with IGD. In the current study, our aim was to investigate cortical morphology and further explore the relationship between the cortical morphology and symptom severity in male youths with IGD. Forty-eight male youths with IGD and 32 age- and education-matched normal controls received magnetic resonance imaging scans. We employed a recently proposed surface-based morphometric approach for the measurement of cortical thickness (CT). We found that youths with IGD showed increased CT in the bilateral insulae and the right inferior temporal gyrus. Moreover, significantly decreased CT were found in several brain areas in youths with IGD, including the bilateral banks of the superior temporal sulci, the right inferior parietal cortex, the right precuneus, the right precentral gyrus, and the left middle temporal gyrus. Additionally, youths with IGD demonstrated a significantly positive correlation between the left insular CT and symptom severity. Our data provide evidence for the finding of abnormal CT in distributed cerebral areas and support the notion that altered structural abnormalities observed in substance addiction are also manifested in IGD. Such information extends current knowledge about IGD-related brain reorganization and could help future efforts in identifying the role of insula in the disorder.
Introduction
According to the authoritative announcement of China Internet Network Information Center, till December 2017, the population of netizens in China has reached 772 million, accounting for about one-fifth of the total population of Internet users worldwide.1 With the rapid popularity of the Internet, the phenomenon of clinical impairments or distress caused by maladaptive use of the Internet has grasped the attention of medical and public health professionals (1–7). Research on maladaptive use of the Internet has become a rapidly evolving field of study (8, 9). In acknowledgment of the studies that have already been published in this field, the Section III of DSM-5 classified “Internet gaming disorder” (IGD) as a condition in need of further research before being officially recognized as an independent clinical disorder (10). As a probable candidate for behavioral addiction, IGD was defined in particular as “persistent and recurrent use of the Internet to engage in games” (11) and has gained massive attention worldwide. Scholars within the field have been motivated to provide empirical evidence for this potential clinical category by applying different study approaches such as epidemiology, psychosociology, and neuroimaging. For example, epidemiological studies have shown that the overall prevalence of IGD ranged from 0.7 to 15.6% in studies of naturalistic populations, with an average percentage of 4.7% over the years (12). In addition, several theoretical models have been proposed for inspiring clear hypotheses on the mechanisms underlying the clinical phenomenon of IGD, which can be useful for the theory-driven development of assessment tools and treatments (12–15).
The technological advancement of neuroimaging, especially non-invasive magnetic resonance imaging (MRI), has made it possible to assess both anatomical and functional brain characteristics of IGD (9). Convergent evidence has indicated that brain structural alterations were associated with individuals with IGD, which suggested an underpinning neuroscientific basis for IGD (8). For example, Han et al. (16) reported increased gray matter volume of the left thalamus in individuals with IGD, and Zhou et al. reported decreased gray matter density of the left cingulate cortex and left insula in individuals with IGD (17). With regard to those structural alterations, there was an influential explanation that the neural mechanisms underlying IGD resemble those of substance addiction (14, 18). Although such behavioral addictions do not involve a chemical intoxicant or substance, study evidence revealed that many aspects of behavioral addiction are similar to those of substance addiction (19, 20). For example, a common neurobiological feature during the resting state (21) and similar impulsivity and executive dysfunctions have been reported between IGD and alcohol use disorder (20). An open question thus is whether these altered structural abnormalities observed in substance addiction also manifest in IGD.
In the past decades, tremendous progress has been made in the techniques and applications of cortical surface morphometry based on structural MRI (22). Previous studies have indicated that surface-based brain mapping may offer advantages over volume-based brain mapping to capture the fine structure of cortical anatomy, since it provides a series of cortical measures that possess anatomical meanings, such as cortical thickness (CT) (22, 23). To the best of our knowledge, so far, very few studies have conducted surface-based brain mapping in the individuals with IGD. Reassuringly, as comparable references, one study has demonstrated the reduction of orbitofrontal CT in male adolescents with Internet addiction (24), and the other revealed a changed CT pattern in late adolescence with online gaming addiction (25). However, both studies were conducted before the publishing of DSM-5, and different criteria were applied throughout those studies. It is our belief that the features of cortical anatomy in IGD are not well known; neither is its association with symptoms of IGD. Therefore, it is necessary to assess the morphological features of IGD using the new DSM-5 approach. In the present study, we used surface-based morphometry (SBM) approaches to examine CT changes of the whole brain in male youths with IGD. According to previous findings derived from studies on IGD (8, 24, 25) and substance addiction (26), we hypothesized that male youths with IGD may have increased CT in the insula. Considering that the insula has been proposed to be crucial for the formation and maintenance of IGD (15), we further speculated that increased insular CT may be associated with symptom severity in male youths with IGD.
Materials and Methods
Participants
All participants were recruited from local universities and the surrounding community via advertisements and word of mouth. Participants were then pre-selected through an online questionnaire and telephone screening. Given the higher prevalence of Internet addiction in males versus females in China (27, 28), only male participants were included. Forty-eight youths who reported Internet gaming as their primary online activity met at least five of the nine DSM-5 criteria for IGD (10). Participant’s Internet addictive behavior was assessed with a Chinese version of Internet Addiction Test (IAT) (29). IAT includes 20 items on a 5-point Likert scale (scored from 1 to 5) indicating the level of Internet usage, with good internal consistency and concurrent validity (30, 31). The higher the score, the greater the problems caused by Internet usage. All IGD subjects satisfied with their score on the IAT more than the proposed cutoff score (i.e., ≥50) (32, 33). Male youths who dissatisfied the proposed criteria for IGD were pre-selected as normal controls (NCs). Among them, 32 participants were determined as NCs based on their score of less than 30 on the IAT. NCs satisfied with fewer than four of the nine criteria for IGD proposed by DSM-5. All participants were right-handed as assessed with the Edinburgh Handedness Inventory (34). A brief structured clinical interview tool, the Mini International Neuropsychiatric Interview (35), was used to screen for several psychiatric disorders. Exclusion criteria for the participants included intracranial pathology, brain injury, neurological disorder, several psychiatric disorders, substance abuse, contraindications for MRI examinations, and excessive head motion. The demographic characteristics of youths with IGD and NCs are summarized in Table 1.
MRI Data Acquisition
Magnetic resonance imaging scans were obtained by using a 3.0 Tesla Magnetom Trio Tim (Siemens Medical System, Erlangen, Germany) at the Department of Medical Imaging, The Affiliated Wuxi People’s Hospital of Nanjing Medical University. Foam pads were used to reduce head motion and scanner noise. Three-dimensional T1-weighted images were acquired by employing a 3D-MPRAGE sequence with the following parameters: time repetition = 2,300 ms, time echo = 2.98 ms, flip angle = 9°, matrix size = 256 × 256, field of view = 256 mm × 256 mm, 160 sagittal slices, slice thickness = 1.2 mm, acquisition voxel size = 1 mm × 1 mm × 1.2 mm, and total acquisition time = 303 s.
MRI Data Processing
To identify cortical alternations in youths with IGD, an SBM was performed using the CAT toolbox2 with the SPM12 software.3 A detailed description of the processing procedure of the CAT toolbox can be found elsewhere.4 In brief, this toolbox uses a fully automated method that allows for measurement of CT and reconstructions of the central surface in one step. It uses a tissue segmentation to estimate the white matter (WM) distance, then projects the local maxima (which is equal to the CT) to other gray matter voxels by using a neighbor relationship described by the WM distance. This projection-based thickness allows the handling of partial volume information, sulcal blurring, and sulcal asymmetries with no need of explicit sulcus reconstruction (36). For statistical analysis of surface measure, the CT images were smoothed with a 15 mm full width-half maximum Gaussian kernel.
Statistical Analysis
To detect statistical significance of group differences in demographic variables between youths with IGD and NCs, the Student’s t-test was used. To determine the cortical changes in youths with IGD, we used an analysis of covariance model with diagnostic group as fixed variable, including age as the confounding covariate. Whole-brain peak-level family wise error corrections with P < 0.05 (two-tailed) were used in all comparisons to ensure the statistical significance. Then, to further delineate the association between the cortical morphology and symptom severity (reflected by total scores of IAT) in youths with IGD and NCs, respectively, a multiple regression model with IAT total scores as the independent variable was used. Since education level and age were significantly correlated within youths with IGD (P < 0.001) and NCs (P < 0.001), the multiple regression model included only age as a confounding covariate. For exploratory analysis, we relaxed the peak-level significance threshold to 0.001 (two-tailed, uncorrected) and the cluster-level significance threshold with cluster-size >100. The scatter plot of the relationship between IAT total scores and the mean values of CT was created using GraphPad Prism.5 Identification of brain regions was determined with the Desikan–Killiany brain atlas (37).
Results
Forty-eight youths with IGD and 32 NCs were analyzed in the present study. No significant differences were detected between youths with IGD and NCs in age and education. Compared with NCs, youths with IGD showed a significant increase in IAT total scores and reported game playing time and scores of DSM-5 criteria (Table 1).
In comparison with NCs, brain areas with significantly increased CT were found in youths with IGD, including the bilateral insulae and the right inferior temporal gyrus (Table 2). Moreover, significantly decreased CTs were found in several brain areas in youths with IGD, including the bilateral banks of the superior temporal sulci (STS), the right inferior parietal cortex, the right precuneus, the right precentral gyrus, and the left middle temporal gyrus (Table 2). In youths with IGD, the regression analysis revealed that the CT values in the left insula were positively correlated with IAT total scores (cluster size = 285, peak coordinate MNIxyz = [−38, −1, −6], t = 4.19, see Figures 2A,B). Compared with NCs, youths with IGD showed significantly increased means of the left insular CT (Figure 2C). No significantly negative correlations were observed between the CT and IAT total scores in youths with IGD. In addition, no significant correlations were observed between the CT and IAT total scores in NCs.
Discussion
The present study used the SBM approach to characterize cortical morphological features in youths with IGD. The primary finding was that youths with IGD had significant CT alterations in distributed cerebral areas, including the insular, parietal, temporal, and frontal cortices. Particularly, youths with IGD showed a significant association between increased insular CT and IGD symptom severity (reflected by IAT total scores). These findings provide new evidence of cortical morphological abnormalities in IGD and highlight a key role played by the insula in the symptom manifestation of this disorder.
Previous studies have demonstrated several specific brain regions associated with IGD, such as the amygdala (38), the insula (39, 40), the precuneus (41), and the middle temporal gyrus (42). In line with the literature, the present study revealed a distributed pattern of CT abnormality in youths with IGD, including the insula, the superior temporal sulcus, the precuneus, the precentral gyrus, and the middle temporal gyrus (Figure 1). Previous studies have shown that Internet game playing was associated with the brain regions responsible for attention and control, impulse control, motor function, emotional regulation, and sensory-motor coordination (14). It is thus conceivable that multiple brain regions have been reported to be probable neural substrates in Internet addictive behavior (38, 39, 43).
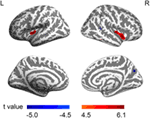
Figure 1. Brain regions with abnormal cortical thickness (CT) in male youths with Internet gaming disorder (IGD). The warm color denotes the brain regions having increased CT, and the cold color denotes the brain regions having decreased CT in youths with IGD. The colored bars show t values. L, left; R, right.

Figure 2. (A) The panel shows the left lateral brain image depicting a correlation between the left insular cortical thickness (CT) and Internet Addiction Test (IAT) total scores in male youths with Internet gaming disorder (IGD). The image was peak-level thresholded at P < 0.001 (two-tailed, uncorrected) and cluster-level thresholded at cluster size > 100 for exploratory analysis. The warm color denotes the positive correlation, and the colored bar shows t values. (B) The scatter plot shows the relationship between means of the left insular CT and IAT total scores in youths with IGD. R2, the coefficient of determination. (C) The histogram illustrates group means of the left insular CT for youths with IGD and normal controls (NCs).*P < 0.05. Error bars reflect the SD.
Among our findings, the abnormality of the insular CT and its association with IGD symptom severity were especially interesting. This finding was in line with the previous structural MRI studies (17, 25, 44), which convergently demonstrated structural changes of the insula in individuals with IGD. Our data were also in line with recent functional MRI studies, one of which reported enhanced activity of the bilateral insulae in individuals with problematic Internet use during a monetary incentive learning task (45). Zhang et al. (39) observed an impaired functional connectivity pattern of the insula in subjects with IGD, and their finding was supported by another research which reported an association between IGD severity and insula-based functional connectivity (40). In terms of function, the insula is believed to play a major role in diverse functions such as multimodal sensory processing (46), social decision making (47), emotional experience (48), and motor control (49). Furthermore, the insula is proposed to integrate internal and external information to raise an awareness of the “global emotional moment” experiences that aid in maintaining a context relevant homeostatic state (50, 51). Neuroimaging and lesion studies have suggested that the insula plays an important role in cigarette smoking behavior (52, 53). Moreover, Tanabe et al. reported that the insula cortex was thicker in substance dependent men (26). According to the proposed tripartite neurocognitive model of IGD (15), the insula should be one of the key components underlying IGD, which maintains the craving for an Internet game. The activity of the insula may enhance the drive to play the Internet game and weaken the inhibitive abilities regarding this action. Thus, our study provided new supporting evidence for this model of IGD and highlighted that involvement of the insula in IGD is similar to those of substance addiction. Such information may help to develop effective intervention strategies. For example, psychopharmacological treatments and psychotherapy targeting the circuits including the insula may be effective in weakening craving in individuals with IGD. On the other hand, our results were comparable with the findings derived from a previous independent SBM study, which reported reduced CT of the left insula in individuals (12 males and 6 females) with online gaming addiction (25). Inconsistent with their hemilateral change pattern of CT in the insula (25), the present study showed increased CT of the bilateral insulae in individuals with IGD. One possible reason for the inconsistent results was the differences in sex composition of the samples. Previous studies have revealed that sex is an important modulator of Internet-related behavior (54). Although the effect of sex on the insular CT in individuals with IGD is still unclear, a recent SBM study demonstrated a diagnosis-by-sex interaction on insular CT in substance-dependent individuals (26). Other possible reasons may be related to the methodology, the sample size, and the heterogeneity of participants. The specific roles of insula in IGD require further investigation in future studies by employing a more comprehensive design. Altogether anatomical and functional abnormalities in the insula were widely implicated in IGD. Our findings extend current knowledge about IGD-related insular cortical morphological characteristics and their associations with clinical symptoms.
Another interesting finding of the current study was the significantly decreased CT in the bilateral banks of STS. The banks of STS, defined as the posterior aspect of STS (37), are involved in the processing of various activities such as recognition of motion and faces and understanding of social cues (55). A recent functional MRI study provided evidence that the posterior STS serves as the hub for the distributed brain network for social perception (56). This suggests that the posterior STS is functionally tightly coupled with other brain circuitries and likely integrates social signals processed by more specialized subsystems (56). Furthermore, experimental studies have indicated the role of STS in both real-life situations and games (57, 58). On the one hand, the gray matter density of the STS was specifically associated with online social network size in healthy participants (58). On the other hand, swear words induced more activation in the STS when compared with negative words in young adolescents with IGD (59). A recent meta-analysis also confirmed that STS has been implicated in “the theory of mind” during human–human interactions (60). Therefore, we believe that our finding of involvement of the STS in IGD is a conceivable consequence, which sheds light on the underlying brain structure in IGD. However, the specific roles of the STS in IGD require additional investigation in future studies by employing a more comprehensive design model considering both the structural and functional requirements.
Several issues need to be further considered. First, we employed a recently proposed projection-based thickness approach (36) for measurement of CT in the present study. Such projection-based thickness approach enables the processing of partial volume information, sulcal blurring, and sulcal asymmetries with no need for explicit sulcus reconstruction either via skeleton or thinning method and may be superior in certain respects to previous approaches (22, 36). Second, whether these abnormalities observed in our data were a consequence or precondition of IGD remains a question yet to be answered. The answer requires further investigation in future studies by employing a more comprehensive design. Third, the Desikan–Killiany brain atlas handles the insula as a whole region. However, functional MRI and histological studies have shown that the insula is not a homogenous cortical region, which could be functionally subdivided into several distinct subregions (61, 62). Future SBM studies are encouraged to employ an atlas with fine subregional structures of insula. In addition, previous studies have demonstrated that behavioral and neural mechanisms of IGD mostly overlap with those of substance use disorders (18). Thus, more cognitive measurements such as rewards, cravings, and memory-related tasks are needed to explain the findings of the present study.
Conclusion
Taken together, our data demonstrated that youths with IGD had significant CT alterations in distributed cerebral areas, including the insular, parietal, temporal, and frontal cortices. Particularly, youths with IGD showed a significantly positive correlation between symptom severity and the left insular CT. This work extends current knowledge about IGD-related cortical morphological features and their associations with clinical symptoms. Such information could help with future efforts to identify the role of the insula in the disorder.
Ethics Statement
This study was carried out in accordance with the recommendations of the Medical Ethics Committee of The Affiliated Wuxi Mental Health Center of Nanjing Medical University with written informed consent from all subjects. All subjects gave written informed consent in accordance with the Declaration of Helsinki. The protocol was approved by the Medical Ethics Committee of The Affiliated Wuxi Mental Health Center of Nanjing Medical University.
Author Contributions
ZZ and LT designed the study. FZ, JL, QT, JW, and LC contributed to the acquisition of the data. LT, SW, and JL analyzed the data, interpreted the results, and drafted the manuscript. All the authors critically reviewed content and approved the final version for publication.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This study was financially supported by the National Natural Science Foundation of China (Nos. 81471354 and 81301148), Special Foundation for Young Medical Talents of Jiangsu Province (No. QNRC2016175), and Special Foundation for Key Medical Talents of Jiangsu Province (joint effort of the provincial and municipal governments). The authors express appreciation to all subjects included in our study for their participation and Ophelia Dandra Bellanfante and Kayris Alanna Foster for English language assistance.
Footnotes
- ^http://www.cnnic.net.cn (Accessed: March 17, 2018).
- ^http://dbm.neuro.uni-jena.de/cat/ (Accessed: March 17, 2018).
- ^http://www.fil.ion.ucl.ac.uk/spm/software/spm12/ (Accessed: March 17, 2018).
- ^http://dbm.neuro.uni-jena.de/cat12/CAT12-Manual.pdf (Accessed: March 17, 2018).
- ^https://www.graphpad.com (Accessed: March 17, 2018).
References
1. Petry NM, O’Brien CP. Internet gaming disorder and the DSM-5. Addiction (2013) 108(7):1186–7. doi:10.1111/add.12162
2. Black DW, Belsare G, Schlosser S. Clinical features, psychiatric comorbidity, and health-related quality of life in persons reporting compulsive computer use behavior. J Clin Psychiatry (1999) 60(12):839–44. doi:10.4088/JCP.v60n1206
3. Young KS. Internet addiction: the emergence of a new clinical disorder. Cyberpsychol Behav (1998) 1(3):237–44. doi:10.1089/cpb.1998.1.237
4. Mak KK, Lai CM, Watanabe H, Kim DI, Bahar N, Ramos M, et al. Epidemiology of internet behaviors and addiction among adolescents in six Asian countries. Cyberpsychol Behav Soc Netw (2014) 17(11):720. doi:10.1089/cyber.2014.0139
5. Zhou Z, Li C, Zhu H. An error-related negativity potential investigation of response monitoring function in individuals with internet addiction disorder. Front Behav Neurosci (2013) 7:131. doi:10.3389/fnbeh.2013.00131
6. Ji SK, Bo HL, Chung SK, Lee H. Correlates of smartphone use and Internet addiction among Korean adolescents: public health perspective. J Behav Addict (2015) 4:23. doi:10.1556/JBA.4.2015.Suppl.1
7. Tran BX, Le TH, Hinh ND, Long HN, Le BN, Nong VM, et al. A study on the influence of internet addiction and online interpersonal influences on health-related quality of life in young Vietnamese. BMC Public Health (2017) 17(1):138. doi:10.1186/s12889-016-3983-z
8. Weinstein AM. An update overview on brain imaging studies of Internet gaming disorder. Front Psychiatry (2017) 8:185. doi:10.3389/fpsyt.2017.00185
9. Zhu Y, Zhang H, Tian M. Molecular and functional imaging of Internet addiction. Biomed Res Int (2015) 2015:378675. doi:10.1155/2015/378675
10. Association AP. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington: American Psychiatric Publishing (2013).
11. Kuss DJ. Internet gaming addiction: current perspectives. Psychol Res Behav Manag (2013) 6:125–37. doi:10.2147/PRBM.S39476
12. Feng W, Ramo DE, Chan SR, Bourgeois JA. Internet gaming disorder: trends in prevalence 1998-2016. Addict Behav (2017) 75:17. doi:10.1016/j.addbeh.2017.06.010
13. Dong G, Potenza MN. A cognitive-behavioral model of Internet gaming disorder: theoretical underpinnings and clinical implications. J Psychiatr Res (2014) 58:7–11. doi:10.1016/j.jpsychires.2014.07.005
14. Weinstein A, Livny A, Weizman A. New developments in brain research of Internet and gaming disorder. Neurosci Biobehav Rev (2017) 75:314–30. doi:10.1016/j.neubiorev.2017.01.040
15. Wei L, Zhang S, Turel O, Bechara A, He Q. A tripartite neurocognitive model of Internet gaming disorder. Front Psychiatry (2017) 8:285. doi:10.3389/fpsyt.2017.00285
16. Han DH, Lyoo IK, Renshaw PF. Differential regional gray matter volumes in patients with on-line game addiction and professional gamers. J Psychiatr Res (2012) 46(4):507–15. doi:10.1016/j.jpsychires.2012.01.004
17. Zhou Y, Lin FC, Du YS, Qin LD, Zhao ZM, Xu JR, et al. Gray matter abnormalities in Internet addiction: a voxel-based morphometry study. Eur J Radiol (2011) 79(1):92–5. doi:10.1016/j.ejrad.2009.10.025
18. Zhang Y, Ndasauka Y, Hou J, Chen J, Yang LZ, Wang Y, et al. Cue-induced behavioral and neural changes among excessive Internet gamers and possible application of cue exposure therapy to Internet gaming disorder. Front Psychol (2016) 7:675. doi:10.3389/fpsyg.2016.00675
19. Lesieur HR, Blume SB. Pathological gambling, eating disorders, and the psychoactive substance use disorders. J Addict Dis (1993) 12(3):89–102. doi:10.1300/J069v12n03_08
20. Zhou Z, Zhu H, Li C, Wang J. Internet addictive individuals share impulsivity and executive dysfunction with alcohol-dependent patients. Front Behav Neurosci (2014) 8:288. doi:10.3389/fnbeh.2014.00288
21. Kim H, Kim YK, Gwak AR, Lim JA, Lee JY, Jung HY, et al. Resting-state regional homogeneity as a biological marker for patients with Internet gaming disorder: a comparison with patients with alcohol use disorder and healthy controls. Prog Neuropsychopharmacol Biol Psychiatry (2015) 60:104–11. doi:10.1016/j.pnpbp.2015.02.004
22. Evans AC. Cortical surface morphometry. Brain Mapp (2015) 2:157–66. doi:10.1016/B978-0-12-397025-1.00210-4
23. Shi J, Wang Y. Surface-based morphometry. Brain Mapp (2015) 1:395–9. doi:10.1016/B978-0-12-397025-1.00310-9
24. Hong SB, Kim JW, Choi EJ, Kim HH, Suh JE, Kim CD, et al. Reduced orbitofrontal cortical thickness in male adolescents with Internet addiction. Behav Brain Funct (2013) 9:11. doi:10.1186/1744-9081-9-11
25. Yuan K, Cheng P, Dong T, Bi Y, Xing L, Yu D, et al. Cortical thickness abnormalities in late adolescence with online gaming addiction. PLoS One (2013) 8(1):e53055. doi:10.1371/journal.pone.0053055
26. Tanabe J, York P, Krmpotich T, Miller D, Dalwani M, Sakai JT, et al. Insula and orbitofrontal cortical morphology in substance dependence is modulated by sex. AJNR Am J Neuroradiol (2013) 34(6):1150–6. doi:10.3174/ajnr.A3347
27. Li Y, Zhang X, Lu F, Zhang Q, Wang Y. Internet addiction among elementary and middle school students in China: a nationally representative sample study. Cyberpsychol Behav Soc Netw (2014) 17(2):111–6. doi:10.1089/cyber.2012.0482
28. Lau JTF, Gross DL, Wu AMS, Cheng KM, Lau MMC. Incidence and predictive factors of Internet addiction among Chinese secondary school students in Hong Kong: a longitudinal study. Soc Psychiatry Psychiatr Epidemiol (2017) 52(6):657–67. doi:10.1007/s00127-017-1356-2
29. Young KS. Caught in the Net: How to Recognize the Signs of Internet Addiction – and a Winning Strategy for Recovery. New York, NY: John Wiley & Sons, Inc (1998). 248 p.
30. Panayides P, Walker MJ. Evaluation of the psychometric properties of the Internet addiction test (IAT) in a sample of cypriot high school students: the Rasch measurement perspective. Eur J Psychol (2012) 8(3):93–9. doi:10.5964/ejop.v8i3.474
31. Widyanto L, McMurran M. The psychometric properties of the Internet addiction test. Cyberpsychol Behav (2004) 7(4):443–50. doi:10.1089/cpb.2004.7.443
32. Dong G, Wu L, Wang Z, Wang Y, Du X, Potenza MN. Diffusion-weighted MRI measures suggest increased white-matter integrity in Internet gaming disorder: evidence from the comparison with recreational Internet game users. Addict Behav (2018) 81:32–8. doi:10.1016/j.addbeh.2018.01.030
33. Dong G, Lin X, Potenza MN. Decreased functional connectivity in an executive control network is related to impaired executive function in Internet gaming disorder. Prog Neuropsychopharmacol Biol Psychiatry (2015) 57:76–85. doi:10.1016/j.pnpbp.2014.10.012
34. Oldfield RC. The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia (1971) 9(1):97–113. doi:10.1016/0028-3932(71)90067-4
35. Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The mini-International neuropsychiatric interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry (1998) 59(Suppl 20):22–33;quiz4–57.
36. Dahnke R, Yotter RA, Gaser C. Cortical thickness and central surface estimation. Neuroimage (2013) 65:336–48. doi:10.1016/j.neuroimage.2012.09.050
37. Desikan RS, Segonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage (2006) 31(3):968–80. doi:10.1016/j.neuroimage.2006.01.021
38. Ko CH, Hsieh TJ, Wang PW, Lin WC, Yen CF, Chen CS, et al. Altered gray matter density and disrupted functional connectivity of the amygdala in adults with Internet gaming disorder. Prog Neuropsychopharmacol Biol Psychiatry (2015) 57:185–92. doi:10.1016/j.pnpbp.2014.11.003
39. Zhang Y, Mei W, Zhang JX, Wu Q, Zhang W. Decreased functional connectivity of insula-based network in young adults with Internet gaming disorder. Exp Brain Res (2016) 234(9):2553–60. doi:10.1007/s00221-016-4659-8
40. Zhang JT, Yao YW, Li CS, Zang YF, Shen ZJ, Liu L, et al. Altered resting-state functional connectivity of the insula in young adults with Internet gaming disorder. Addict Biol (2016) 21(3):743–51. doi:10.1111/adb.12247
41. Wang H, Jin C, Yuan K, Shakir TM, Mao C, Niu X, et al. The alteration of gray matter volume and cognitive control in adolescents with internet gaming disorder. Front Behav Neurosci (2015) 9:64. doi:10.3389/fnbeh.2015.00064
42. Dong G, Potenza MN. Risk-taking and risky decision-making in Internet gaming disorder: implications regarding online gaming in the setting of negative consequences. J Psychiatr Res (2016) 73:1–8. doi:10.1016/j.jpsychires.2015.11.011
43. Kuhn S, Gallinat J. Brains online: structural and functional correlates of habitual Internet use. Addict Biol (2015) 20(2):415–22. doi:10.1111/adb.12128
44. Lin X, Dong G, Wang Q, Du X. Abnormal gray matter and white matter volume in ‘Internet gaming addicts’. Addict Behav (2015) 40:137–43. doi:10.1016/j.addbeh.2014.09.010
45. Yoon H, Kim SA, Ahn HM, Kim SH. Altered neural activity in the anterior and posterior insula in individuals with problematic Internet use. Eur Addict Res (2015) 21(6):307–14. doi:10.1159/000377627
46. Bushara KO, Grafman J, Hallett M. Neural correlates of auditory-visual stimulus onset asynchrony detection. J Neurosci (2001) 21(1):300–4.
47. Quarto T, Blasi G, Maddalena C, Viscanti G, Lanciano T, Soleti E, et al. Association between ability emotional intelligence and left insula during social judgment of facial emotions. PLoS One (2016) 11(2):e0148621. doi:10.1371/journal.pone.0148621
48. Phan KL, Wager T, Taylor SF, Liberzon I. Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in PET and fMRI. Neuroimage (2002) 16(2):331–48. doi:10.1006/nimg.2002.1087
49. Anderson TJ, Jenkins IH, Brooks DJ, Hawken MB, Frackowiak RS, Kennard C. Cortical control of saccades and fixation in man. A PET study. Brain (1994) 117(Pt 5):1073–84.
50. Jones CL, Ward J, Critchley HD. The neuropsychological impact of insular cortex lesions. J Neurol Neurosurg Psychiatry (2010) 81(6):611–8. doi:10.1136/jnnp.2009.193672
51. Craig AD. How do you feel – now? The anterior insula and human awareness. Nat Rev Neurosci (2009) 10(1):59–70. doi:10.1038/nrn2555
52. Naqvi NH, Rudrauf D, Damasio H, Bechara A. Damage to the insula disrupts addiction to cigarette smoking. Science (2007) 315(5811):531–4. doi:10.1126/science.1135926
53. Morales AM, Ghahremani D, Kohno M, Hellemann GS, London ED. Cigarette exposure, dependence, and craving are related to insula thickness in young adult smokers. Neuropsychopharmacology (2014) 39(8):1816–22. doi:10.1038/npp.2014.48
54. Vyjayanthi S, Makharam S, Afraz M, Gajrekar S. Gender differences in the prevalence and features of internet addiction among Indian college students. Medica Innovatica (2014) 3:5196–201.
55. Wyk BC, Hudac CM, Carter EJ, Sobel DM, Pelphrey KA. Action understanding in the superior temporal sulcus region. Psychol Sci (2009) 20(6):771–7. doi:10.1111/j.1467-9280.2009.02359.x
56. Lahnakoski JM, Glerean E, Salmi J, Jaaskelainen IP, Sams M, Hari R, et al. Naturalistic FMRI mapping reveals superior temporal sulcus as the hub for the distributed brain network for social perception. Front Hum Neurosci (2012) 6:233. doi:10.3389/fnhum.2012.00233
57. Haruno M, Kawato M. Activity in the superior temporal sulcus highlights learning competence in an interaction game. J Neurosci (2009) 29(14):4542–7. doi:10.1523/JNEUROSCI.2707-08.2009
58. Kanai R, Bahrami B, Roylance R, Rees G. Online social network size is reflected in human brain structure. Proc Biol Sci (2012) 279(1732):1327–34. doi:10.1098/rspb.2011.1959
59. Chun JW, Choi J, Cho H, Lee SK, Kim DJ. Dysfunction of the frontolimbic region during swear word processing in young adolescents with Internet gaming disorder. Transl Psychiatry (2015) 5:e624. doi:10.1038/tp.2015.106
60. Schurz M, Tholen MG, Perner J, Mars RB, Sallet J. Specifying the brain anatomy underlying temporo-parietal junction activations for theory of mind: a review using probabilistic atlases from different imaging modalities. Hum Brain Mapp (2017) 38(9):4788–805. doi:10.1002/hbm.23675
61. Cauda F, Dagata F, Sacco K, Duca S, Geminiani G, Vercelli A. Functional connectivity of the insula in the resting brain. Neuroimage (2011) 55(1):8. doi:10.1016/j.neuroimage.2010.11.049
62. Kurth F, Eickhoff SB, Schleicher A, Hoemke L, Zilles K, Amunts K. Cytoarchitecture and probabilistic maps of the human posterior insular cortex. Cereb Cortex (2010) 20(6):1448–61. doi:10.1093/cercor/bhp208
Keywords: cortical thickness, insula, Internet gaming disorder, surface-based morphometry, symptom severity
Citation: Wang S, Liu J, Tian L, Chen L, Wang J, Tang Q, Zhang F and Zhou Z (2018) Increased Insular Cortical Thickness Associated With Symptom Severity in Male Youths With Internet Gaming Disorder: A Surface-Based Morphometric Study. Front. Psychiatry 9:99. doi: 10.3389/fpsyt.2018.00099
Received: 10 January 2018; Accepted: 13 March 2018;
Published: 03 April 2018
Edited by:
Marc N. Potenza, Yale University, United States
Reviewed by:
Aviv M. Weinstein, Ariel University, Israel
Yuan-Wei Yao, Beijing Normal University, China
Copyright: © 2018 Wang, Liu, Tian, Chen, Wang, Tang, Zhang and Zhou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.

